Category: Respiratory
-
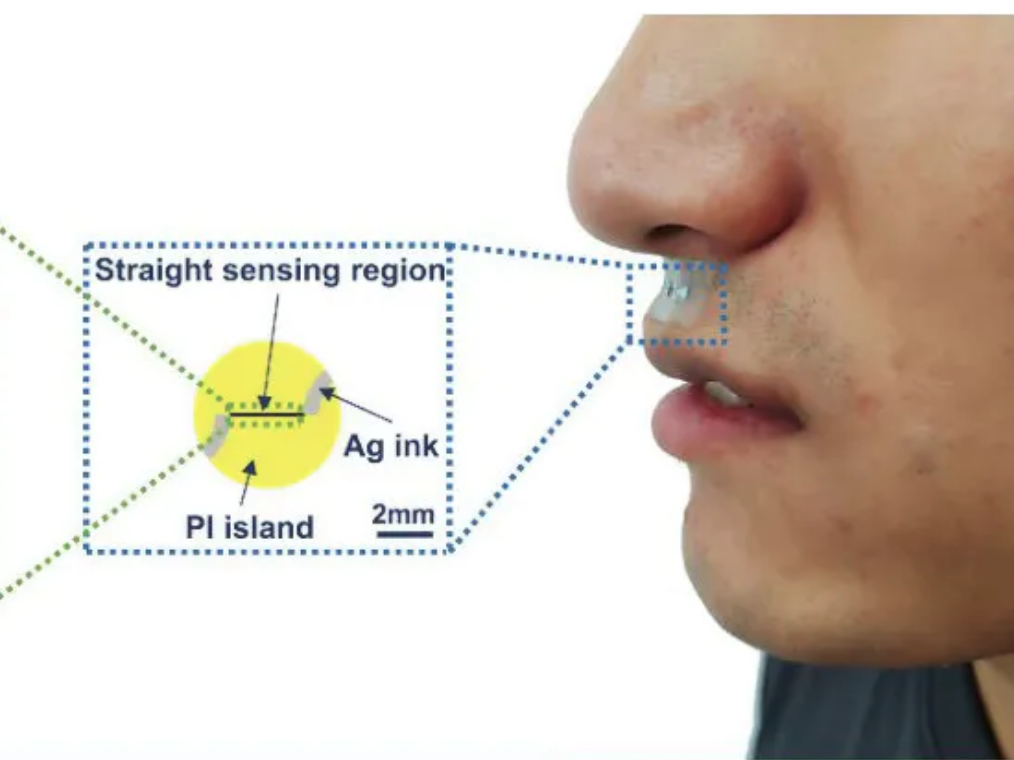
Small sticker-sensor continuously analyzes breath for broad health monitoring
Heibei University, Tianjin Hospital, Beihang University, and Penn State researchers have developed an under nose-worn, stretchable, skin-friendly, waterproof sensor to analyze breath for health monitoring. It could be use for multiple-condition screening, asthma and COPD management, or environmental hazard sensing, among other applications. The functional gas sensor, in a moisture-resistant membrane, can operate in humid…
-
3D printed, custom fitted airway stents
Cleveland Clinic physician Tom Gildea used CT scans and visualization software to develop 3D printed silicone stents used to open the airways of patients with tumors, inflammation, trauma or other masses. The technology has now been approved by the FDA. Standard airway stents come in a limited number of sizes and shapes and are generally…
-

Biodegradable piezoelectric sensor monitors lungs, brain
UConn’s Thanh Duc Nguyen has developed a biodegradable pressure sensor to monitor chronic lung disease, swelling of the brain, and other health issues. It is small and flexible and designed to replace existing, potentially toxic, implantable pressure sensors. Those sensors must be removed, subjecting patients to another invasive procedure, prolonging recovery, and increasing infection risk.…
-

Lung function analyzed via phone, from anywhere
Mayank Goel and University of Washington colleagues have developed SpiroCall, a system that measures lung function by analyzing a caller’s voice via smartphone, landline or payphone microphone. An algorithm uses the phone microphone as an uncalibrated pressure sensor. Captured audio is converted into an estimate of the flow-rate of air exiting from a patient’s mouth.…
-
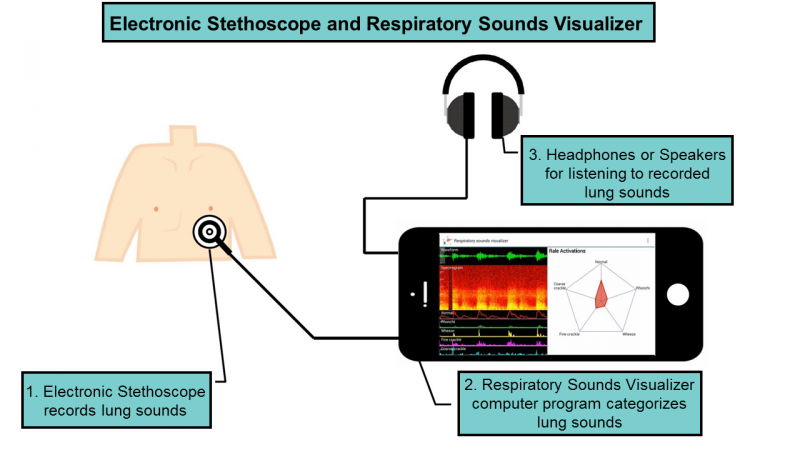
Stethoscope software analyzes lung sounds
Hiroshima University and Fukushima Medical University researchers have created software and an electronic stethoscope to classify lung sounds into five common diagnostic categories. Currently, doctors listening to heart and lung sounds on a stethoscope need to overcome background noise and recognize multiple irregularities. The system will be able to “hear” what a doctor might miss, and automatically…
-

Respiratory motion system for improved lung tumor imaging
U of T professor Shouyi Wang has developed a mathematical model based, personalized respiratory motion system for more precise lung tumor imaging. Respiratory gating, or a patient’s motion breath-by-breath, is monitored, and the data is used to focus a radiology beam on the target when the chest cavity is relaxed. This is the stage that…
-

Ingestible sensor continuously monitors heart, breathing rates
MIT researchers are developing ingestible sensors that measure heart and breathing rates from within the gastrointestinal tract using sound waves. This type of sensor could make it easier to assess trauma patients, monitor soldiers in battle, perform long-term evaluation of patients with chronic illnesses, or improve training for professional and amateur athletes, the researchers say.…
-
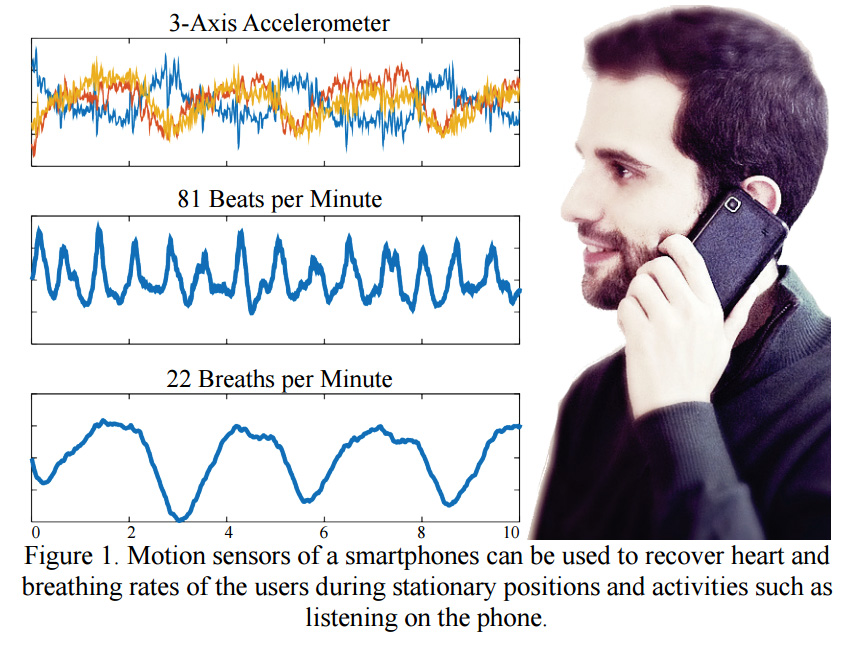
Phone tracks health with out wearable sensors
Javier Hernandez Rivera of Rosalind Picard‘s Affective Computing Group at MIT is developing a health monitoring phone that does not require a wearable. BioPhone derives biological signals from a phone’s accelerometer, which the team says captures small body movements that result from one’s heart beating and chest rising and falling. Hernandez said that BioPhone is meant…
-

Phone sensor detects asthma attacks, triggers
Wing by Sparo Labs is a crowdfunded smartphone attachment that detects early signs of asthma attacks. Its sensor works with a companion app to measure FEV1 (how much air one can exhale in one second) and Peak Flow (how fast one can exhale). The company claims that data it collects over time by can allow users…
-

Piezoelectric sensor car seat monitors respiration, heart rate
Faurecia‘s “Active Welness” car seat monitors respiration and heart rate with embedded piezoelectric sensors. The goal is to detect driver stress or alertness. When low energy is detected, the seat responds with specific massage patterns and air flow through the ventilation system. The non-contact sensors were developed by Hoana Medical. Combined with advanced algorithms and signal processing, Faurecia…
-
Phone sensors measure oxygen saturation with out pulse oximeter
MoveSense allows oxygen saturation to be monitored by phone sensors with what its developers describe as medical accuracy. A mobile phone must be carried in one’s pocket, and no pulse oximeter is required. The technology was developed by Bruce Schatz at the University of Illinois. In a study, patients wore pulse oximeters (for comparison) and carried phones…
-
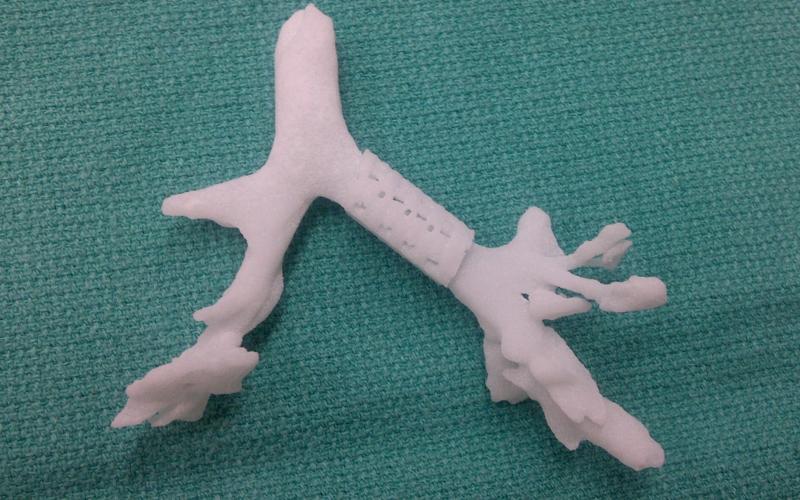
3D printed airway splints restore breathing
At the University of Michigan, three children under 2 with tracheobronchomalacia had 3D printed devices implanted to open their airways and restore their breathing. Professors Glenn Green and Scott Hollister were able to create and implant customized tracheal splints for each patient. The device was created directly from CT scans of their tracheas, integrating an image-based…