Category: 3-D Printing
-
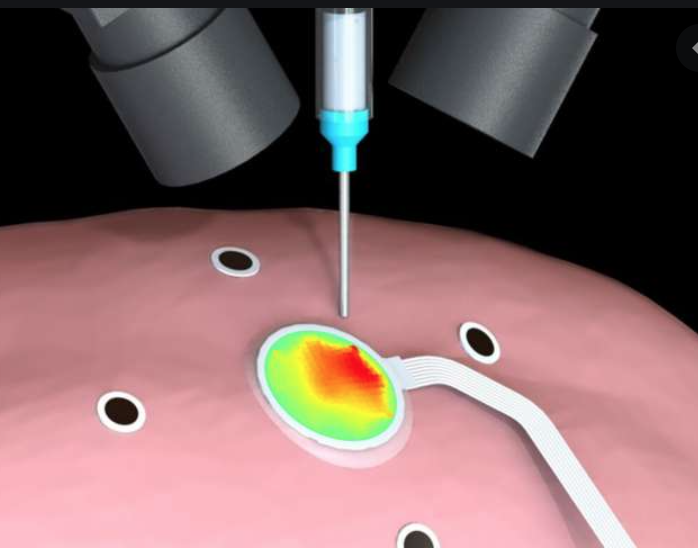
Sensors 3D printed directly on lungs, heart could be used with surgical robots to diagnose, monitor disease
Michael McAlpine and University of Minnesota colleagues used 3D printing and motion capture technology to print electronic sensors directly on organs that are expanding and contracting, such as the heart and lungs. This could be used to diagnose and monitor the lungs of patients with COVID-19. This builds on the team’s technique which enabled the…
-
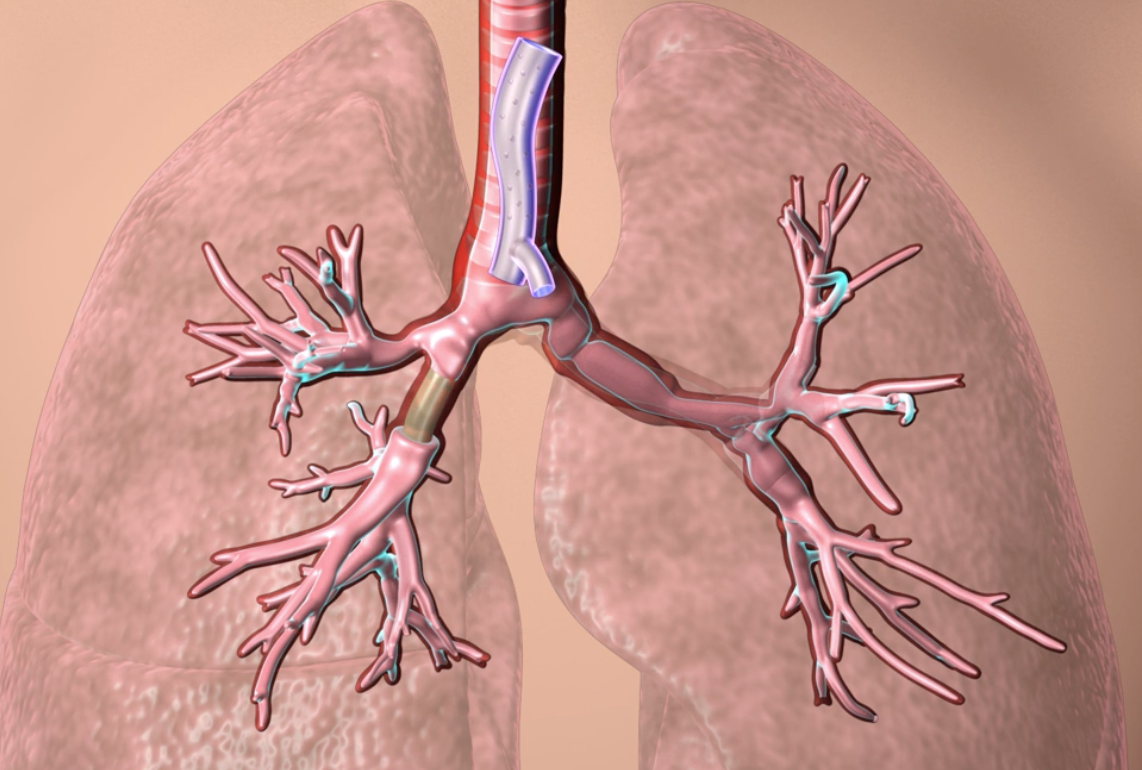
3D printed, custom fitted airway stents
Cleveland Clinic physician Tom Gildea used CT scans and visualization software to develop 3D printed silicone stents used to open the airways of patients with tumors, inflammation, trauma or other masses. The technology has now been approved by the FDA. Standard airway stents come in a limited number of sizes and shapes and are generally…
-
3D-printed, remote-controlled lab on chip for quicker, more accurate monitoring
Imperial College’s Martyn Boutelle has developed a 3D printed, remote-controlled Lab on a Chip for real-time monitoring with improved personal care. Previous ‘Lab on a Chip’ devices have required large external support systems. Sensor deterioration over time inhibited their clinical effectiveness. The device can monitor chemical fluctuations, giving quicker and more accurate results, and potentially…
-

3D printed skin, with blood vessels
Pankaj Karande and Rensselaer Polytechnic Institute colleagues have developed 3D printed living skin, with blood vessels. which could integrate with host cells in grafts. Until now, a significant barrier to integration has been the absence of a functioning vascular system. Karande previously made two types of living human cells into “bio-inks,” and print them into a skin-like…
-
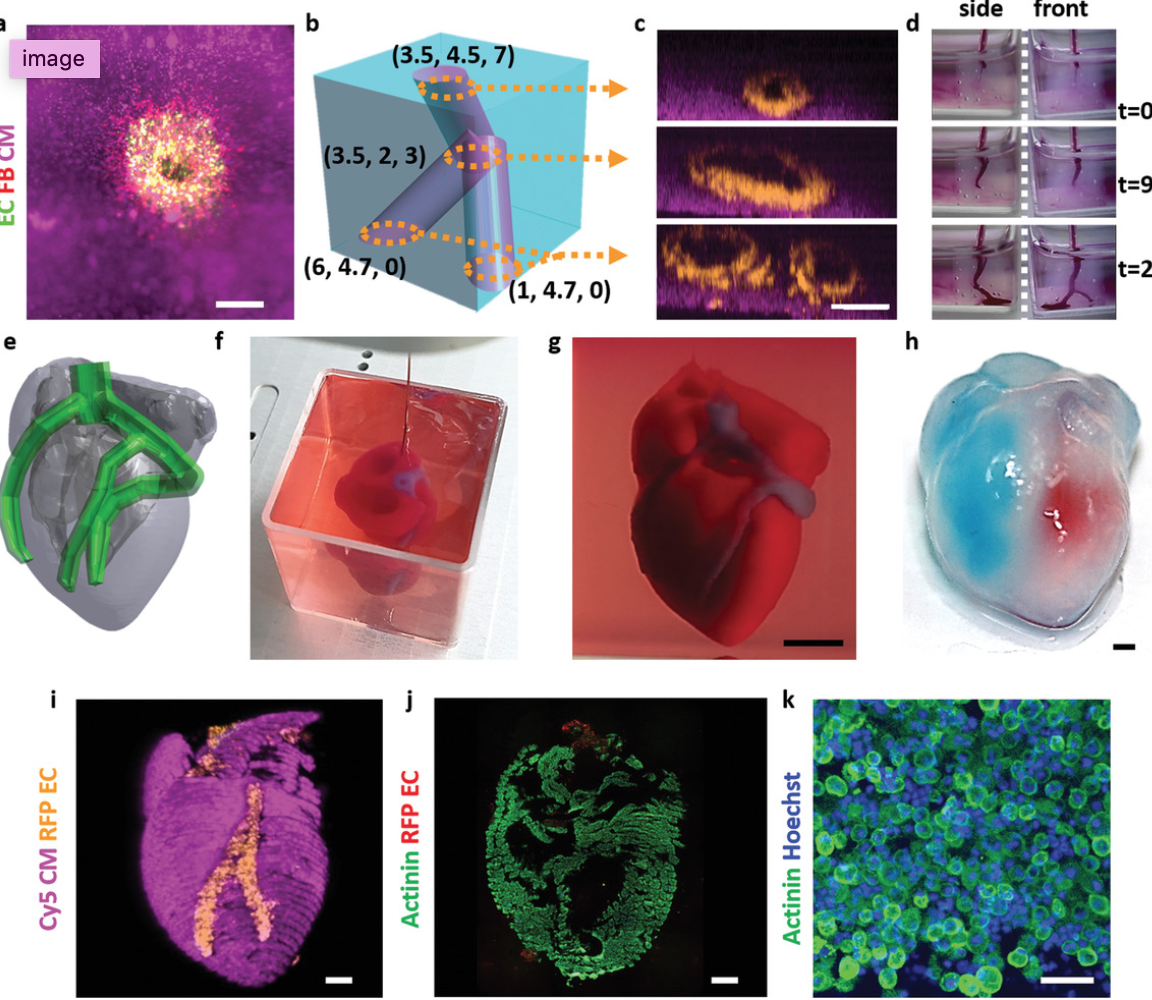
3D printed, vascularized heart, using patient’s cell, biological materials
Tel Aviv University professor Tal Dvir has printed a 3D vascularized engineered heart, including cells, blood vessels, ventricles and chambers, using a patient’s own cell and biological materials. A biopsy of fatty tissue was taken from patients. Cellular and a-cellular materials were separated. While the cells were reprogrammed to become pluripotent stem cells, the extracellular…
-
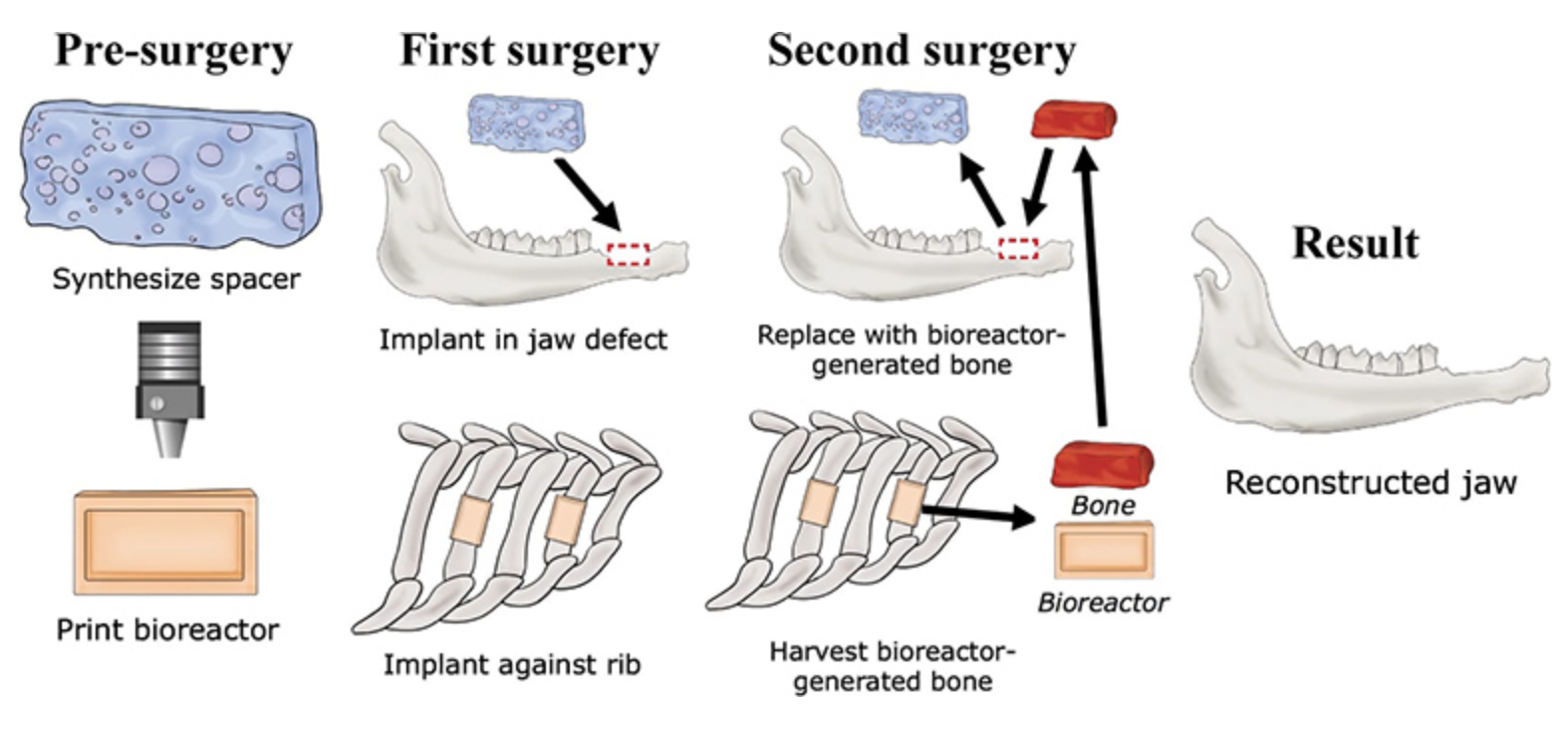
3D printed bioreactor-grown bone for craniofacial surgery
Antonios Mikos, Alexander Tatara, and Rice colleagues are using a 3D printed mold, attached to a rib, to grow live bones to repair craniofacial injuries. Stem cells and blood vessels from the rib infiltrate scaffold material and replace it with natural, custom-fit bone. Current reconstruction methods use a patient’s own bone graft tissues, harvested from the lower…
-

3D-printed, bluetooth-controlled ingestible capsule delivers drugs, senses environment
MIT’s Bob Langer and Giovanni Traverso have developed a 3D-printed, wirelessly-controlled, ingestible capsule that can deliver drugs, sense environmental conditions, or both. It can reside in the stomach for a month. Data is sent to a user’s phone, and instructions from the phone are sent to the device. The sensor could also communicate with other wearable…
-

Proof of concept 3D printed cornea
Newcastle University’s Che Connon has developed proof-of-concept research that could lead to a 3D printed cornea. Stem cells from a healthy donor cornea were mixed with alginate and collagen to create a printable bio-ink. A 3D printer extruded the bio-ink in concentric circles to form the shape of a human cornea in less then 10 minutes. The…
-
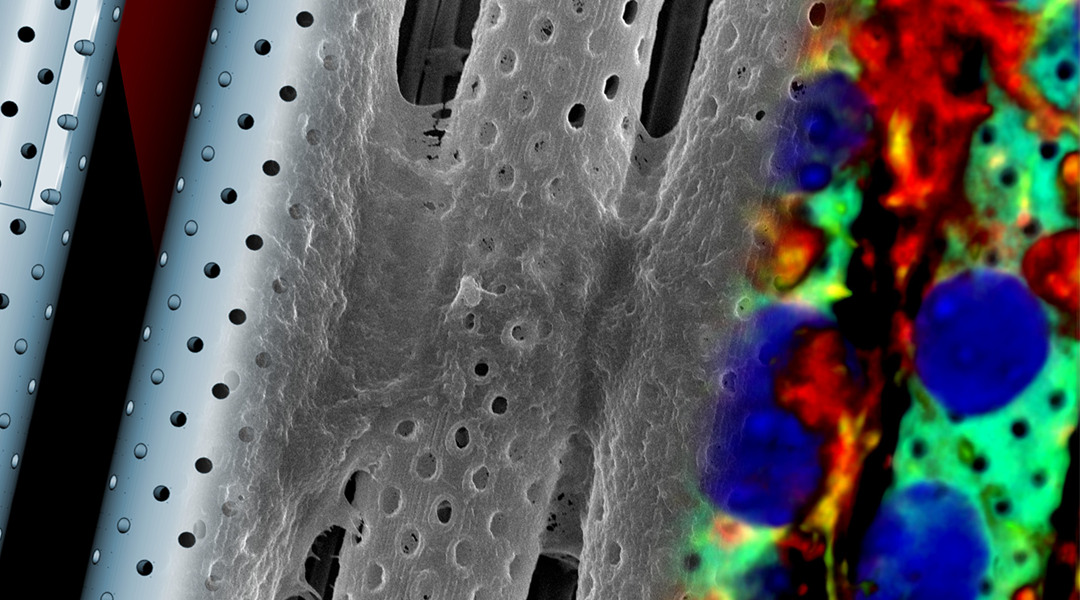
3D, real-scale blood brain barrier model used to study new therapeutics
Gianni Ciofani of ITT Pisa has created a device that reproduces a 1:1 scale model of the blood-brain barrier. The combination of 3D printed artificial and biological components will allow the study of new therapeutic strategies to overcome the blood-brain barrier and treat brain diseases, including tumors, Alzheimers, and multiple sclerosis. A laser that scans…
-

3D-bioprinted human skin can replace animal testing, potentially be used in burns
José Luis Jorcano at Universidad Carlos III de Madrid has developed a 3D bioprinter capable of replicating the structure of skin. The human-like skin that is produced includes an epidermal layer that protects against the environment, and a collagen-producing dermis that provides elasticity and strength. The bioink material contains human plasma, and primary human fibroblasts…
-
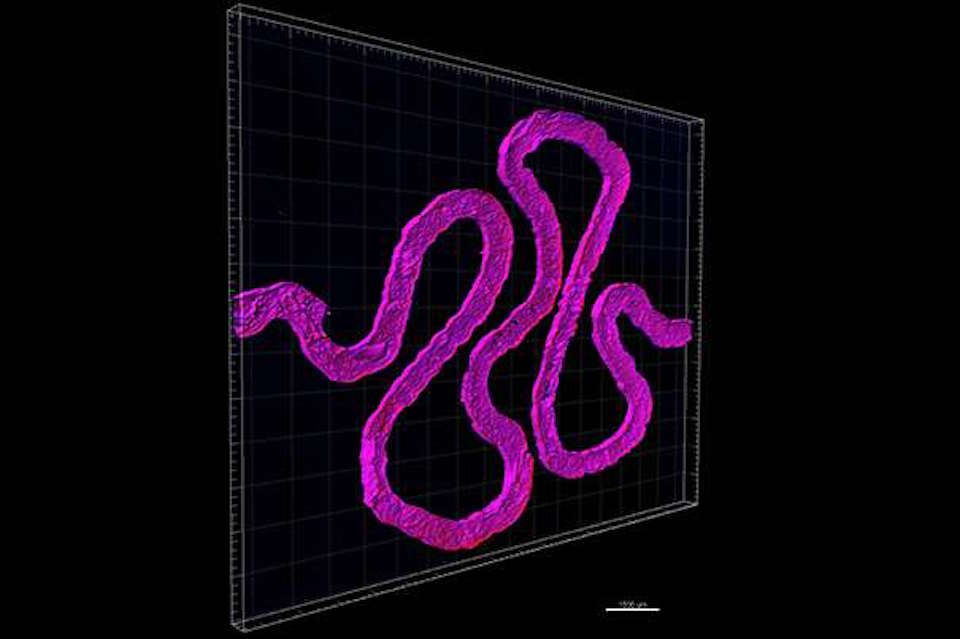
3D printed renal architecture
Harvard’s Jennifer Lewis and Roche’s Annie Moisan have used 3D printing to fabricate a small but critical subunit of a kidney. The renal architecture contains living epithelial cells. Earlier bioprinting approaches were adapted to form thick tissues. A 3D-printed silicone gasket was used to cast an engineered extracellular matrix as a base layer. “Fugitive ink” was…
-
3D printed gel model replicates brain folding mechanism
L. Mahadevan and Harvard colleagues have used 3D printing to replicate a folding human brain. The goal is to understand how brain folds are related to disease. While many molecular processes determine cellular events, the study shows that what ultimately causes the brain to fold is a mechanical instability associated with buckling. A 3D gel model…