Category: Ultrasound
-

Wearable ultrasound to detect early breast cancer
MIT’s Canan Dagdeviren has developed a flexible ultrasound patch that can be attached to a bra, obtaining ultrasound images with resolution comparable to medical imaging centers, and used repeatedly. Interval cancers, which develop between regularly scheduled mammograms, account for 20 – 30 percent of all breast cancers, and tend to be more aggressive. The goal…
-

Implanted ultrasound allows powerful chemotherapy drugs to cross the blood brain barrier
Adam Sonaband and Northwestern colleages used a skull-implantable ultrasound device to open the blood-brain barrier and repeatedly permeate critical regions of the human brain, to deliver intravenous chemotherapy to glioblastoma patients. This is the first study to successfully quantify the effect of ultrasound-based blood-brain barrier opening on the concentrations of chemotherapy in the human brain.…
-

Wearable cardiac ultrasound continuously monitors heart structure, function for 24 hours
UCSD professor Sheng Xu has developed a wearable ultrasound device that can continuously monitor and assess the structure and function of the human heart for 24 hours, during normal daily activity. This could eliminate the need for highly trained technicians and bulky devices. Signs of cardiac diseases are transient and unpredictable, and imaging can detect…
-

Study: Focused ultrasound reduced essential tremor symptoms for 3 years
In a recent study, Casey Halpern and colleagues used ultrasound to relieve symptoms of essential tremor, for up to three years. The treatment is used when medication does not work. 76 people with an average age of 71 who had essential tremor for an average of 17 years were studied. 56 received focused ultrasound thalamotomy, and…
-
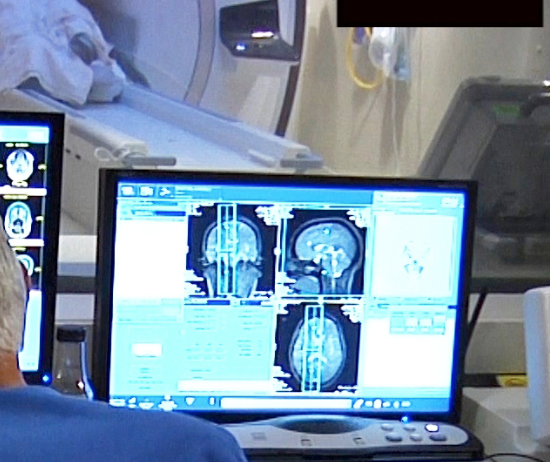
Focused ultrasound thalamotomy in Parkinson’s Disease
UVA’s Scott Sperling and Jeff Elias, who already used focused ultrasound to treat essential tremor, have just published the results of a small study showing the efficacy of the technology in Parkinson’s Disease. The sound waves were shown to interrupt brain circuits responsible for the uncontrollable shaking associated with the disease. The researchers claim that their study…
-
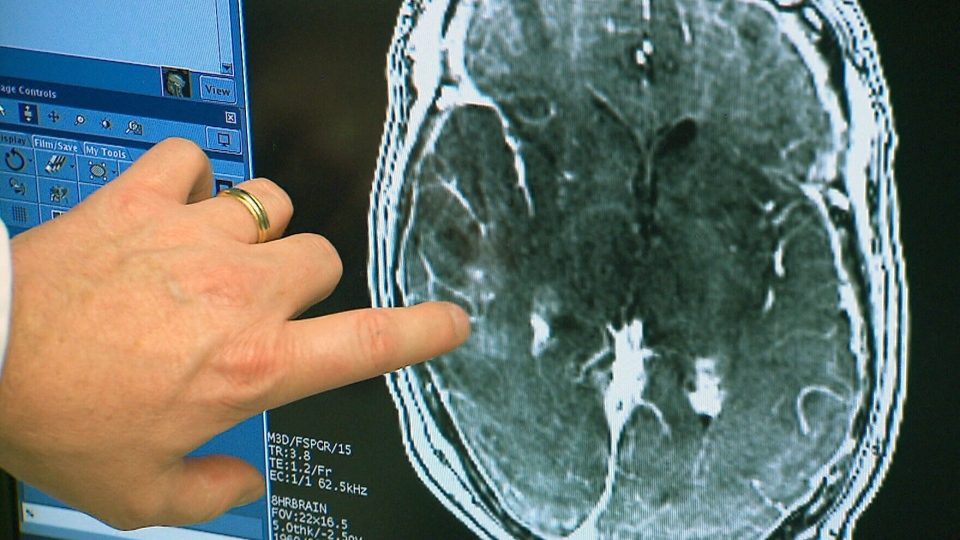
Ultrasound penetrates blood-brain barrier to treat brain tumor
Todd Mainprize at Sunnybrook Hospital has, for the first time, delivered chemotherapy directly to a brain tumor, by breaking through the blood-brain barrier using tightly focused ultrasound. The patient’s bloodstream was infused with a chemotherapy drug, as well as microscopic bubbles, which are smaller than red blood cells and can pass freely through blood. MRI-guided, low intensity sound…
-

Sonogenetics: Neuron stimulation via ultrasound
Salk‘s Sreekanth Chalasani‘s “sonogenetics” technique uses ultrasound to stimulate individual brain cells. A nature paper describes the technology as tested on worms. The goal is noninvasive stimulation of specific cell types or individual neurons in humans, with out using implanted electrodes or fiber-optic cables. Current optogenetics therapies rely on inserting light-sensitive channel proteins into neurons. When hit…
-
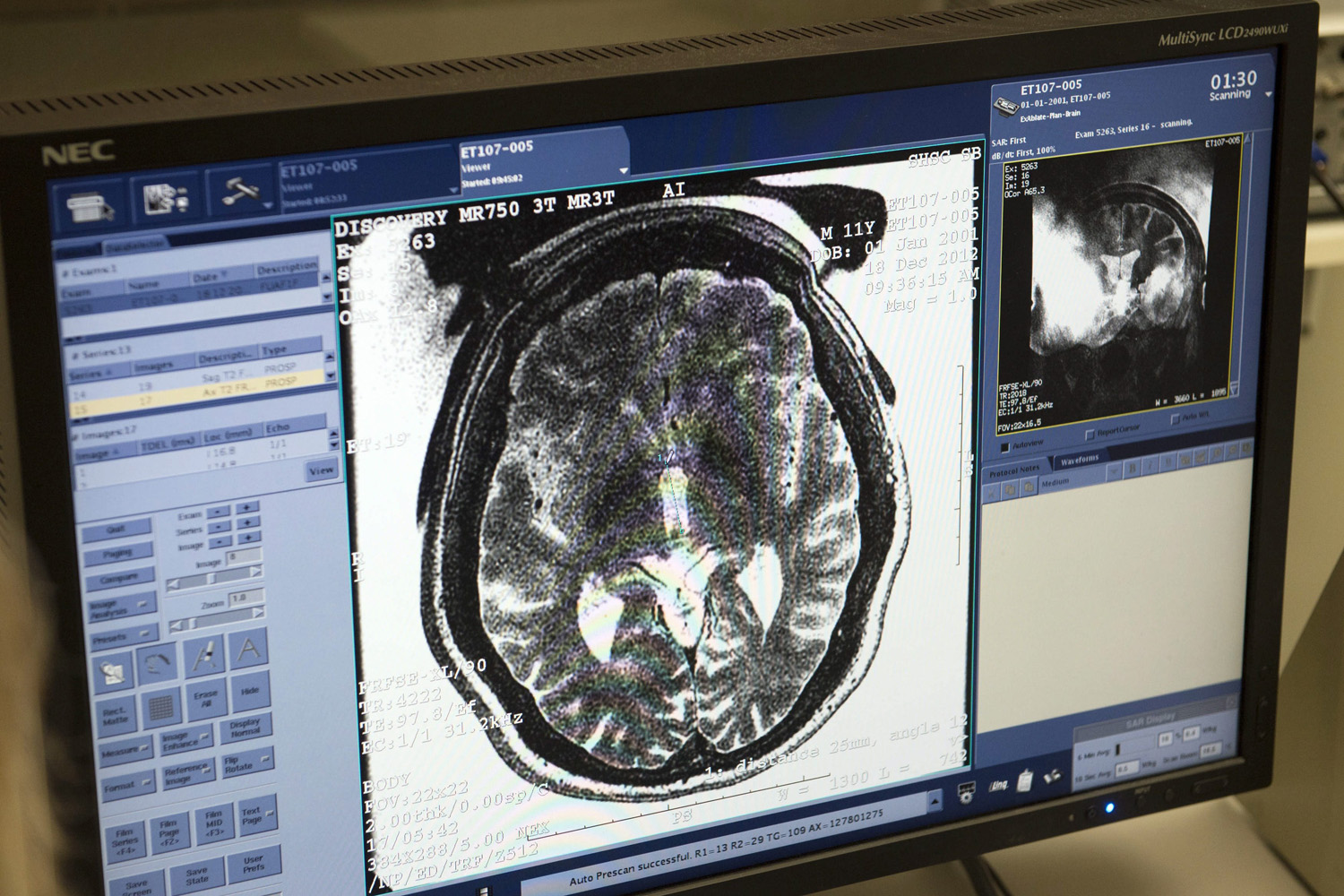
Ultrasound targets deep brain region, helps Parkinson’s symptoms
University of Maryland researchers are using MRI-guided focused ultrasound on the globus pallidus to treat Parkinson’s symptoms. The ExAblate Neuro system was developed by Israel’s Insightec. The treatment is non-invasive, as it does not require a cut, but its ultrasound impacts a deep region of the brain, which is not with out risk. Currently, drugs and…
-
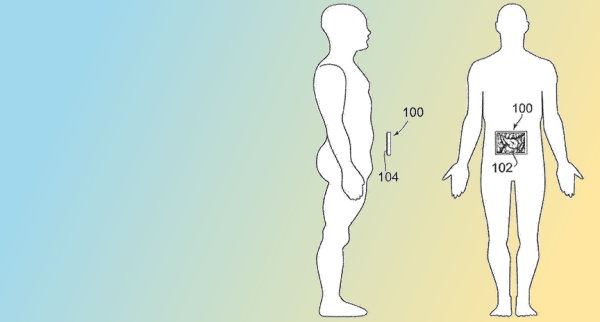
Cheap, fast, precise, hand held ultrasound
Butterfly Network‘s Jonathan Rothberg wants to make a “super-low-cost version of a $6 million (ultrasound) machine, to make it 1,000 times cheaper, 1,000 times faster, and a hundred times more precise.” This will depend on software and extensive AI image research to extract key features to automate diagnoses. Butterfly’s patent applications describe compact, hand held…
-

Ultrasound improves virtual “touch”
Ultrahaptics uses ultrasound waves to make one feel as if he/she is touching virtual objects and surfaces with bare hands. It’s creator, a University of Bristol graduate student, claims that it improves upon touch-free interfaces such as Kinect and Leap Motion by reflecting air pressure waves off the hand to create different sensations for each fingertip. Applications could include interacting with moving…
-
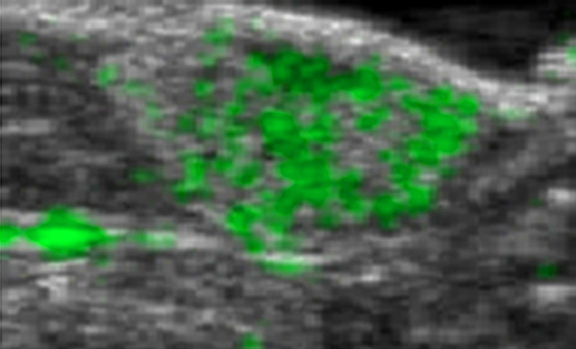
Ultrasound combined with contrasting agent for radiation-free tumor detection
http://www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0086642 University of North Carolina Professor Nancy Klauber-Demore has improved the resolution and tumor-detecting ability of ultrasound scans. Combining ultrasound with a contrast agent composed of tiny bubbles that pair with an antibody that many cancer cells produce at higher levels than do normal cells, Klauber-Demore was able to visualize lesions created by angiosarcoma. By binding to…
-
Ultrasound stimulation enhanced sensory performance in human brain
http://www.nature.com/neuro/journal/vaop/ncurrent/full/nn.3620.html Virginia Tech Carilion Research Institute scientists, led by Professor William Tyler, have demonstrated that ultrasound directed to a specific region of the brain can boost performance in sensory discrimination. This is the first example of low-intensity, transcranial-focused ultrasound modulating human brain activity to enhance perception. The scientists delivered focused ultrasound to an area of the cerebral…